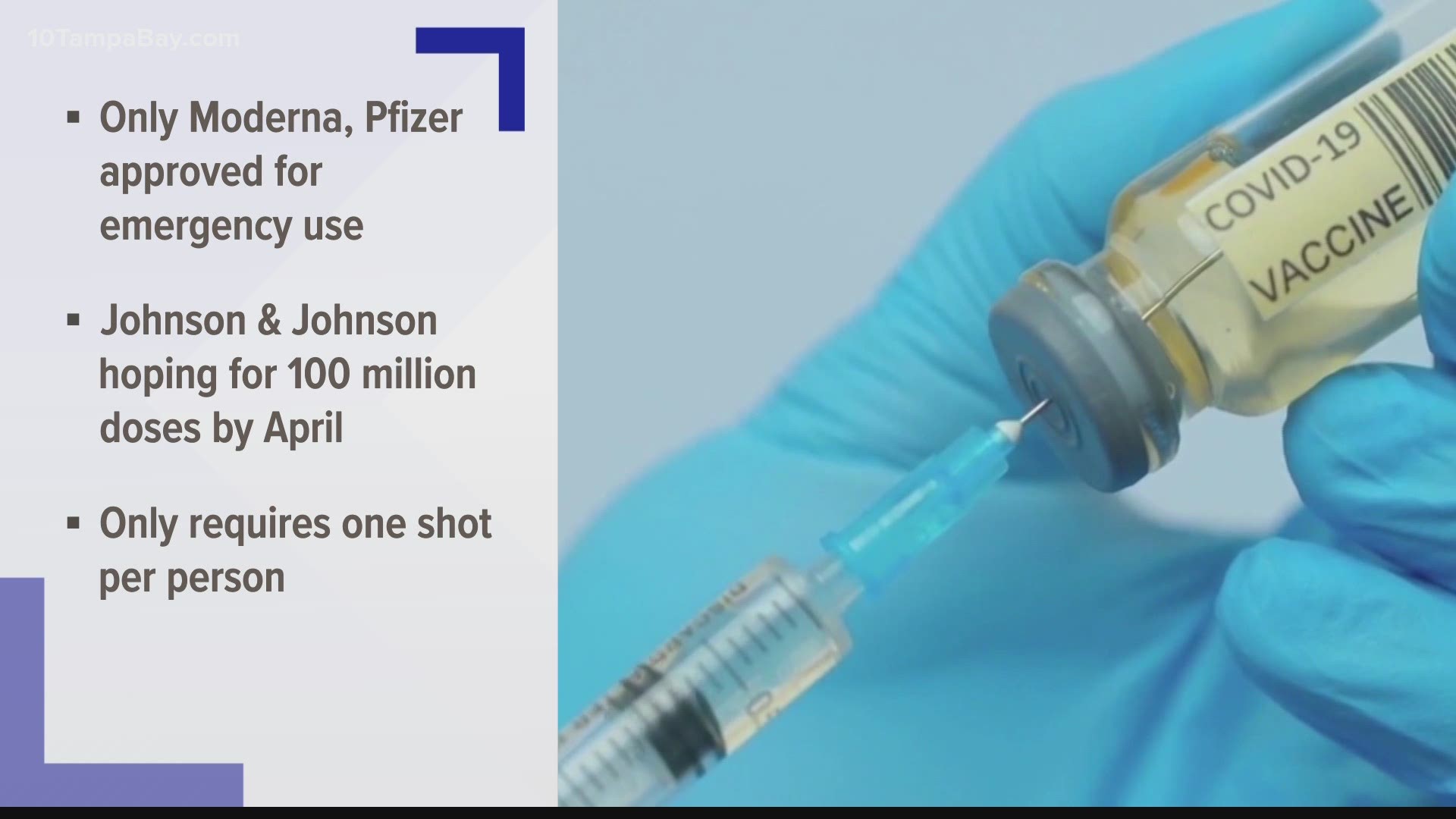
A board member for Johnson & Johnson says the company could have 100 million COVID-19 vaccines ready for Americans by April "if the clinical trial works out."
Dr. Mark McClellan, a former Food and Drug Administration commissioner, told CNBC on Thursday that Johnson & Johnson is "going all out with its production" both in the U.S. and other parts of the world.
Dr. Anthony Fauci, the director of the National Institute of Allergy and Infectious Diseases, said there should be enough data on the Johnson & Johnson vaccine to begin analysis in the next week or two, according to CNBC.
President Joe Biden has set a goal of 100 million coronavirus vaccinations within the first 100 days of his administration. If Johnson & Johnson is able to get its vaccine out by April, that would be on the tail-end of that 100 days.
The Centers for Disease Control and Prevention says the U.S. is currently averaging about 883,000 vaccinations per day, according to CNBC.